A COVID-19 therapy that can help high-risk patients stay healthy enough to avoid a hospital visit is being launched in hospitals in South Carolina, although the drug is not approved and its benefits may be limited.
The drug, called bamlanivimab, has not received full approval from the U.S. Food and Drug Administration. It has authorization for emergency use, which means that it went through an agile process configured at the beginning of the pandemic to verify safety and benefits.
Given the limited supply and a rushed approval process, the drug is not available on a first-come, first-served basis. It is intended for people who test positive for COVID-19 and who are at high risk of developing a serious case due to age or other conditions. Healthcare professionals are deciding which patients are the best candidates.
Trident Health in North Charleston says it has the highest dose available among hospitals in the area, with 150 available. He started offering treatment this week.

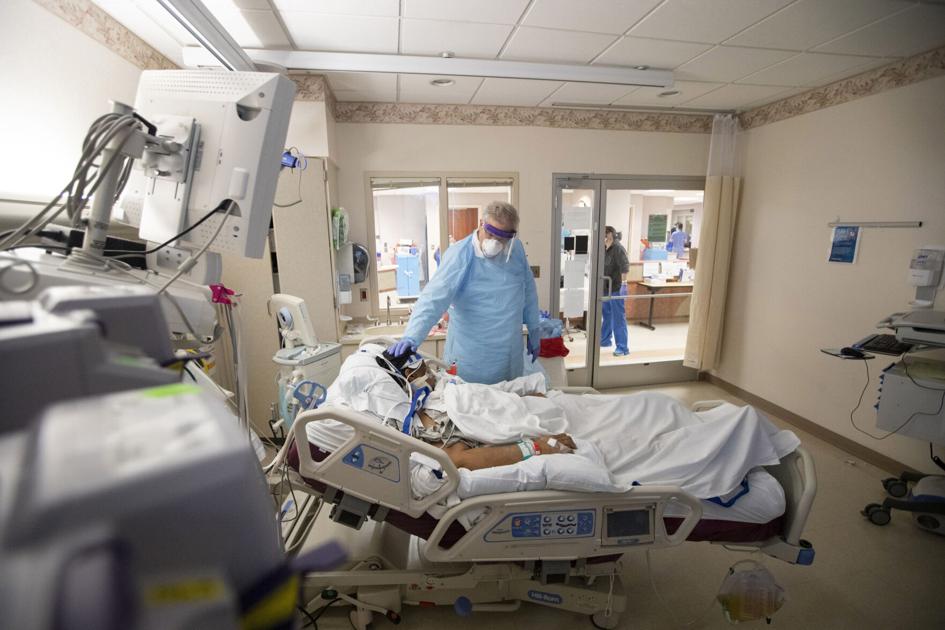
A shot of the novel bamlanivimab therapy COVID-19 in an infusion room at Trident Medical Center in North Charleston. Trident Health offered to receive the maximum possible doses and began administering the drug to qualified patients this week. Mary Katherine Wildeman / Staff
Dr. Lee Biggs, medical director at Trident Health, said the drug is built with antibodies from recovered COVID-19 patients. He said the drug’s benefits are “extremely positive” in keeping patients out of hospital beds.
“In COVID-19, it is gold to be able to stem the progress of the disease,” he said.
South Carolina Medical University said Tuesday it was the first in the state to offer therapy. He initially had 20 doses, giving them all on Wednesday, and a hospital spokeswoman said he had received another 28 doses.
It only goes to patients with MUSC who meet specific criteria. Because of limited supply, MUSC does not accept any external references for the drug, the spokeswoman said.
Conditions that can qualify a patient for the drug include obesity, diabetes, kidney disease and old age, to name a few.

The Roper St. Francis system had access to 50 bottles, but distributed only a few.
Dr. Kent Stock, an infectious disease specialist at Roper St. Francis, said the drug is controversial among healthcare providers, given the limited research completed before the drug was distributed to the general public.
The FDA granted its emergency authorization to the manufacturer, Eli Lilly and Co., on November 9. It works by preventing the virus from replicating within the body.
But Stock said the clinical trial showed that the drug did not effectively reduce patients’ viral load. Even so, it brought other benefits to some patients, including reduced risk of hospitalization. The side effects of the study were limited, although each hospital has to monitor patients carefully.
When it came to Roper St. Francis’ final decision to administer the therapy, Stock said the doctors hoped it would offer enough benefits to be worth it.
“The simple truth is that the medical community is trying to find the best way to use this, based on the information we have,” said Stock. “There is probably a segment of the population that can benefit from this, and if we focus our efforts there, we can probably add value.”

The SC Department of Health and Environmental Control is responsible for distributing the drug to interested hospitals, said Biggs. Trident Health asked for the maximum possible doses. A East Cooper Medical Center spokeswoman confirmed that Mount Pleasant Hospital is not managing it.
Tidelands Health on the Grand Strand received 30 doses in the first distribution, said Gayle Resetar, head of operations.
“I would much rather have someone recover at home with their family than be in a hospital bed,” she said.
Some hospitals are choosing not to use the therapy because it should be administered as an infusion, which presents logistical challenges. Tidelands Health has reserved space for COVID-19 patients at one of its infusion centers in Murrells Inlet, said Resetar.
For Leeann Bauch, a MUSC employee who recently hired COVID-19, bamlanivimab was the right choice.

Bauch, a nurse who runs a dozen departments at MUSC, was diagnosed with tracheomalacia in February. The condition occurs when the trachea does not have the correct shape, making breathing difficult; it is common in children, but not in adults in their 60s like Bauch. It also means that having COVID-19 is particularly risky.
After Bauch obtained her positive COVID-19 test recently, it was only a day after she was receiving the infusion of the new therapy at the MUSC clinic at Citadel Mall. She said she had a headache, but she felt she was back to normal a few days later – no trip to the hospital was necessary.
“I would say it did exactly what it was designed to do,” she said.

Reach Mary Katherine Wildeman at 843-607-4312. Follow her on Twitter @mkwildeman.